
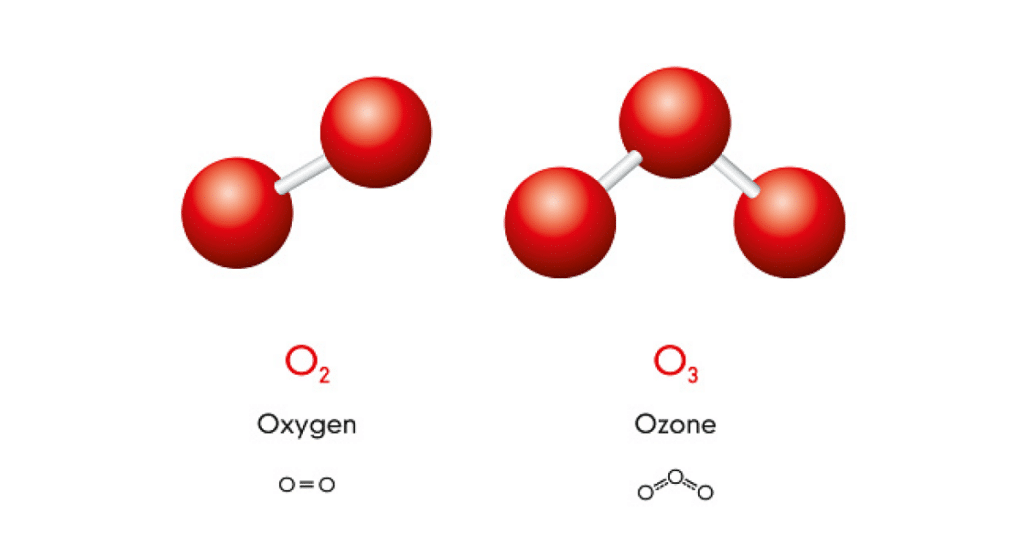
Ozone is a colorless gas that has an odor similar to the smell of the air after a major thunderstorm. The oxygen we breathe (O2) is made up of two oxygen atoms that naturally bind to each other and are very stable. Ozone gas (O3) has three oxygen atoms which consist of a stable pair of (O2) atoms and a third very unstable atom. It is this third unstable atom that gives ozone its exceptional power.
Ozone is a fast-acting powerful germicidal agent, far more powerful than peroxide, chlorine, chloramines, or chlorine dioxide, and capable of rapidly neutralizing bacteria, viruses, fungi, mildew, and other organisms, from water all without toxic compounds or leaving harmful residues. Ozone is also an extremely powerful oxidizer, far more efficient than many chemical processes currently used. Ozone will precipitate out of solution many minerals, heavy metals, and organic or chemical compounds with a short reaction time.

Our Ozone built systems have positive impacts with all types of farming – but especially with extremely sensitive plants like Mixed-Greens and Cannabis. Whether outdoor farming, greenhouses, or hydroponic, all farms recognize the immediate benefits of using Ozone produced water.
One of many benefits using ozonated water is the increased levels of dissolved oxygen (DO), which alone, will significantly improve the farms disinfection process by creating an aerobic environment for the plants resulting in the control or elimination of pathogens – reduction in crop losses – increased plant health and yields.


EXCELLENT
Incredible Difference in Our Crops
We can count on them more than any other contractor or service provider we know!
Thank you Watson Well for saving our well!
Don, his sons, and employees are all very knowledgeable and professional and did our projects at a very reasonable cost.
Thank you so much for the excellent response and care we have received.
I can only offer my highest praise for Don and his employees; thus I rank them 10 out of 10.

CA License #947596C-57 | C-36


Watson Well specializes in Pumps, Water Systems, Water Treatment, and Plumbing for Residential, Commercial, and Agricultural customers. We service Northern California customers from our Redding, Eureka, and Chico locations.
Complete the form below to request an appointment from our team. We will contact you shortly.